More service, more security
Guided by our conviction "More service, more security", we give our best every day as one of the leading metrological service providers. For our customers, our employees and the future of our company.
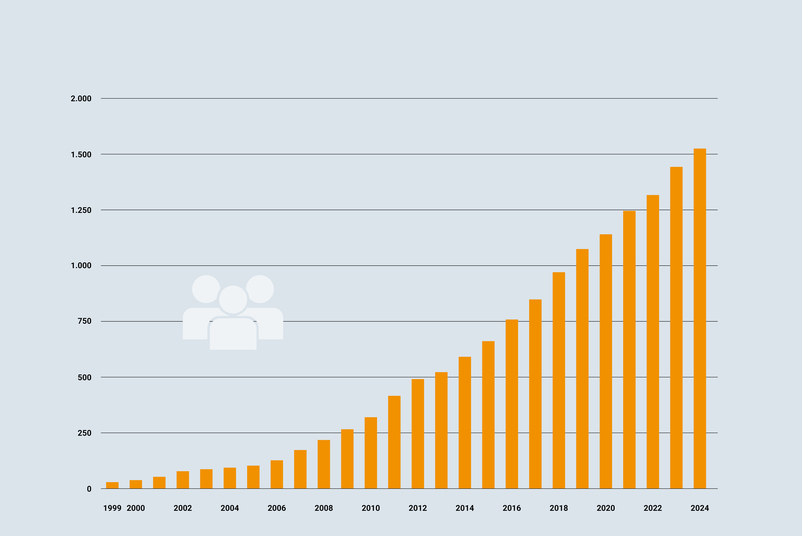
More than 1,500 employees work for you throughout Europe. Quality, reliability, and low measurement uncertainties are the cornerstones of our work. Every single employee - from the apprentice to the managing director - contributes to the success of the company with a large part of his or her own responsibility. With the Testo Industrial Services team, you can be sure that all your measurement, calibration and quality assurance requirements will be fully met.
As a reliable partner at your side
We offer a comprehensive portfolio of services and are at your disposal for questions concerning calibration, test equipment management, qualification and validation. As a subsidiary of Testo SE & Co.KGaA one of the world's largest manufacturers of measuring instruments, we have more than 60 years of experience in the field of measurement technology and quality assurance. With a high degree of self-motivation and cooperative teamwork we successfully shape our common future.

Career & jobs
A unique team spirit and many exclusive employee benefits make us unmistakable.

Services
For our customers we always offer the suitable and demand-oriented solution.

Quality promise
You can always trust in the quality of our services.
Testo Group
We are part of a strong group of companies and give our customers security.
Our company history
The history of Testo Industrial Services GmbH began in 1999 with the founding of the independent subsidiary Testo CAL. Originating from a department of Testo SE & Co. KGaA, Testo CAL started with a handful of employees and the motivation to offer customer oriented, metrological services.
With a spirit of innovation and the constant expansion of its portfolio, the Testo team has always been one step ahead. The doer mentality and motivation of each and every employee are the drivers of the company's success.
1999
- Establishment of the calibration centre in Ingolstadt
- Foundation of Testo CAL GmbH
- DKD accreditation for surface temperature
2000
- Accreditation of the DKD Laboratory Printing
- First FDA/GMP-compliant qualification of pharmaceutical plants
2001
- Establishment of the calibration centre near Marburg
2002
- Takeover of the DKD laboratory for electrical measurands in Munich
- Accreditation for DKD printing on site
2003
- Accreditation of electrical measurement quantities in the HF range
- Accreditation for DKD temperature on site
- Change of name to Testo Industrial Services GmbH
2004
- Relocation of the company management to Kirchzarten near Freiburg i.Br.
- Foundation of the subsidiary Testo industrial services empresarial S.A., Barcelona/Spain
2005
- Accreditation of dimensional measurands
- PRIMAS - Online portal for test equipment management
2006
- Relocation of the laboratories of Testo industrial services from Lenzkirch to Kirchzarten
- Accreditation for electrical measurands on site
- DKD extension: Noble thermocouples with an MU of 0.8°C
2007
- Foundation of the subsidiary Testo industrial services AG, Egg/Switzerland
- New laboratory at the Munich site: Relocation to Karlsfeld near Munich
- DKD extensions:
DC resistance (MU from 0.5 ppm)
DC voltage (MU from 0.5 ppm)
Humidity dew point down to -35° Tpt
2008
- Foundation of the subsidiary Testo Industrial Services S.A.R.L. Forbach/France
- Takeover of Cameco Messtechnik in Pforzheim by Testo industrial services, associated DKD accreditations at the Pforzheim site for electrical and dimensional measurands
- Testo industrial services AG, Egg/Switzerland:
- Accreditations for electrical and dimensional measurands
- Extension of the functions in the online portal for gauge management PRIMAS online
- Awarded "Service Provider of the Year 2008" in the category Service Innovation by the Ministry of Economics Baden-Württemberg
2009
- DKD accreditation at the Kirchzarten site for volumetric flow, mass flow and the electrical measurands DC voltage, DC current intensity, DC current resistance, DC current power, AC voltage, AC current intensity, capacitance, inductance, frequency, time interval, oscilloscopes, temperature simulation
- Foundation of the Calibration Center Essen
- PRIMAS exchange: PRIMAS exchange enables simple, automated data exchange between customer specific MES/CAQ systems and the calibration service provider.
- PNEUMATOR pressure calibrator and gauge: Development and market launch of the device in desk-top format, which is characterized by its wide stationary as well as mobile application range.
- Accreditation of dimensional measurands at the Karlsfeld site
- Accreditation of dimensional measurands at the Essen site
- Accreditation for temperature at the CH/Egg site
- Development of the service product MODUS: Modular Quality Services by Testo Industrial Services - Offers the unique, customer-specific selection of validation, qualification and calibration services in the pharmaceutical environment.
- Development of a validation standard for the Testo SAVERIS system
2010
- Start of construction of the third phase at the Kirchzarten site
- Changeover from DKD to DAkkS
- DKD/DAkkS accreditation for the measurand acceleration at the Kirchzarten site
- Once again among the best two in the service competition of the Ministry of Economics Baden-Württemberg
- Establishment of the new laboratory for the calibration of HF measurement quantities
- Qualification/calibration project at Novartis Vaccines (250 plant qualifications and over 1400 measuring points)
2011
- Accreditation in the HF area
- New accreditation of all measurands by DAkkS; minimized measurement uncertainties
- Pipette calibration
- Move to a new building in Essen
- Completion and ceremonial opening of the third construction phase in Kirchzarten with a new training center, social rooms for employees and a bistro. The expansion has created space for 100 additional workplaces.
- Acquisition of the Testo laboratory in Cabrils, Spain
- New logistics center at the Hanover site
- Expansion of the service portfolio: Pipette calibration at your site
- Relocation of the Marburg Regio-Center to a larger location
- Successful completion of GMP upgrade at B.Braun in Sempach/CH
2012
- New Regio-Center Rhine-Main-Neckar in Ludwigshafen
- New calibration mobile
- Relaunch PRIMAS online
- New Sales Center in Madrid, Spain
- Extension of primary laboratory temperature by normal resistance and fixed point calibration
- HV calibration up to 50kV
- New logistics center in Ingolstadt
- Certification of Testo Industrial Services Switzerland according to ISO 9001
- Integration of Testo Industrial Services France into the group of companies
- Relocation of Testo Industrial Services Spain and France to new buildings
2013
- New service center in Winsen near Hamburg
- Site expansion Kirchzarten
- Extension of the service portfolio to include purity measurements of compressed air and process gases
- Headcount: 400 mark in Germany and 500 mark in Europe exceeded
2014
- 20 years of DAkkS accreditation
- Foundation of Testo Industrial Services GmbH in Austria
- Unique in the world: Measuring robot for length measurement calibration
- Establishment of the GMP training centre in Kirchzarten
- Service Provider of the Year 2014: Testo Industrial Services among the top 3
- ISO certificate with the number 1,000,000 was written
2015
- Relocation Service-Center Munich from Karlsfeld to Dachau
- Accreditation for flow measurement at the Dachau site
- Headcount: 500 mark in Germany and 600 mark in Europe exceeded
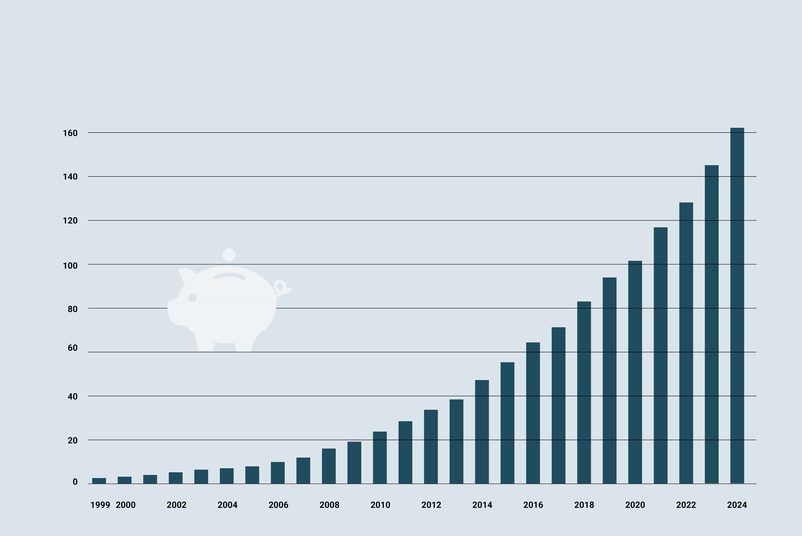
- Most successful year since the company was founded - growth >20% / EUR 6.708 million
2016
- Successful reaccreditation - new DAkkS certificate for five sites and around 200 calibration procedures
- Successful recertification ISO 9001:2008
- Headcount: 600 mark in Germany and 700 mark in Europe exceeded
- Jürgen Hinn becomes fourth member of the board at Testo - new composition of the management Testo Industrial Services
- Groundbreaking ceremony for the next building extension in Kirchzarten
- First in-house typing campaign with the Freiburg stem cell registry: More than 100 employees participate
- ISO certificate with the number 2.000.000 was generated
2017
- Expansion of calibration portfolio: Measured variable Acoustics
- TMS GmbH becomes Testo Industrial Services - New accredited service centre in Hamburg
- New Service: High quality repairs of electrical measuring equipment
- Successful accreditation as a test laboratory for cleanroom qualification measurements
- Completion of extension building 3. Tower at the Kirchzarten Service Centre
- First successful blood donation campaign at Testo Industrial Services
2018
- Introduction of PRIMAS mobile app
- New logistics center in Kulmbach (Northern Bavaria)
- Opening of the service center in Mörfelden-Walldorf (Rhine-Main area)
- Relocation Hamburg
- Successful assessment of our test laboratory by the DAkkS
- Extensions in the scope of accreditation
- Accreditation of acoustic calibrations
- Successful certification in the field of environment and energy according to DIN EN ISO 14001:2015 and DIN EN ISO 50001:2011
- New standard version: Certification according to DIN EN ISO 9001:2015 (instead of 2008)
2019
- 20 years Testo Industrial Services - Anniversary year with many highlights (link to chronicle)
- 20 years - 20 social actions
- Groundbreaking ceremony Calibration Factory
- Changeover to DIN EN ISO/IEC 17025:2018
- TIS welcomes its 1,000th employee
- Relocation of Testo Industrial Services Austria to new premises
- TIS author team writes chapter "Calibration" for the GMP Advisor
- Accreditation certificate for the Service Center in Mörfelden-Walldorf
- 250 engineers & technicians in the technical field service
2020
- Foundation of the subsidiary, Testo Industrial Services Ltd in Great Britain
- Generation change - Christian Sander takes over his father's work as Head of Metrology
- Introduction of the Testo Industrial Services Portal - all information about the order process can be accessed online
- Introduction PRIMAS connect: Enables automated access to all data that can be called up online in PRIMAS in the data system of Testo Industrial Services
- Awarded as FOCUS-Business Top Employer 2020 in the category Services
2021
- Takeover of Wiepro Messtechnik GmbH, measurement services for 3D coordinate measuring technology in Villingen-Schwenningen
- Opening and certification of the Calbration Factory
- Introduction of PRIMAS validated: validatable test equipment management system for companies with GxP compliance requirements
- First-time implementation of webinars, digital seminars and a virtual customer day
- Thomas Richter celebrates his 40th company anniversary at Testo
2022
- Foundation of the subsidiary Testo Industrial Services sp. z o.o. in Poland
- Accredited provider for proficiency testing/ring comparisons according to DIN EN ISO/IEC 17043:2010
- Validation of PAS and Cleanux: GMP-compliant automation systems for calibration and cleanroom measurement
- Water test bench: calibration with water up to 5,000 l/min
- Automated evaluation of climate distribution measurements: Development and introduction of the "Mappux" software
- Expansion of our service centers in Essen and Dachau
- 15th anniversary of Testo Industrial Services AG in Switzerland
- Number of employees in the GxPS division of Germany reached 200 FTE
- Award for Job Motor 2021 in the category: Best concepts for employee recruitment and retention (more than 200 employees)
2023
- Relocation of TIS Poland to a new building in Warsaw
- Accreditation as the first German laboratory for flue gas
- Acquisition of PWT and opening of our new service center in Heidenheim
- Digital calibration certificates rolled out to UK customers helping to reduce our impact on the environment
- Expansion of the portfolio to include test process management services
- New laboratories in France for dimensional, electrical, pressure and multi-parameter calibrations
- Opening of new site in Madrid, Spain
- Investment in the parameter Torque in the UK allowing the laboratory to calibrate torque wrenches and drivers according to DIN EN ISO 6789
- For the first time more than 50 employees in Austria
- Arnt König takes over the position as Managing Director of Testo Industrial Services Switzerland
2024
- Acquisition of the the calibration business TER Calibration Ltd in the Noth West of England in May 2024 to strengthen the position of TIS UK
- Foundation of Testo Industrial Services Kft in Budapest / Hungary with Márk Farkas as Managing Director
- TIS celebrates many anniversaries: 10 years TIS Austria, 20 years TIS Spain, 25 years TIS Germany!
- TIS Poland enlarges to 10 employees
- TIS welcomes the 1,500th employee and the 250th trainee to the group of companies
- Integration of our state-of-the-art coordinate measuring technology into the Kirchzarten service centre and expansion of the building in Gewerbestraße Kirchzarten by more than 100 sqm
- TIS Germany takes over the recalibration of manual measuring technology for Mahr GmbH
- Relocation and enlargement of the Service Center Lyon from Saint-Priest to Genas, France
- ISO 9001 – TIS Austria as well as two branches of TIS France are now also part of the certification
- Successful SCS re-accreditation according to ISO/IEC 17025:2017 of TIS Switzerland that proves ongoing commitment to quality and excellence