Knowledge from the field of medical technology
The knowledge area medical technology gives you an overview of the most important changes of the update of DIN EN ISO 13485. In addition, you will find information on the topics of validation of manual processes and supplier management.
Here you will find answers to the following questions:
- What changes did the update of DIN EN ISO 13485 bring?
- Do manual processes in medical technology need to be validated?
- What is a design history file and what needs to be considered when creating one?
- How can an efficient supplier management be structured for the quality assurance of outsourced processes?
What changes did the update of DIN EN ISO 13485 bring?
Excerpt from the most important changes of the new DIN EN ISO 13485:
- Additional regulatory requirements in the areas of validation, verification, design and development
- Risk management is given greater consideration within the quality management system
- Requirement for software validation is made more explicit
Do manual processes in medical technology need to be validated?
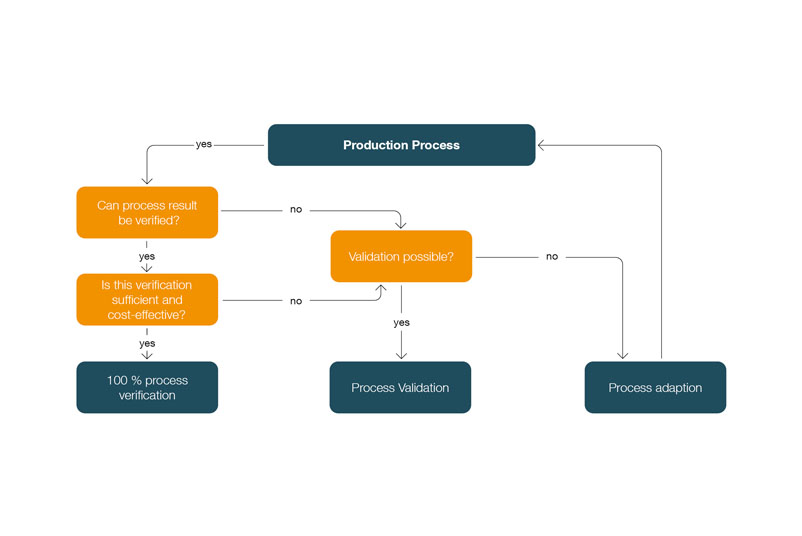
According to the regulatory requirements of ISO standard 13485 and the Quality System Regulations of the FDA, processes whose process results are not (sufficiently) verified or cannot be verified require validation - regardless of whether the process involves manual activities. Manual processes must therefore be considered in exactly the same way as automated processes.
Validation of manual processes in medical technology
This results in the following decision tree for determining whether a process should be validated:
What is a design history file and what needs to be considered when creating one?
The Design History File (DHF) represents a record of the development history and development results of a medical device. Specifically to be documented include:
- Intended Use of the Product
- Requirements and their detailing with specifications
- Design description and operation
- Construction drawings
- Test plans for design verification
- Test plans for design validation
Learn more about the topic in one of our success stories:
How can an efficient supplier management be structured for the quality assurance of outsourced processes?
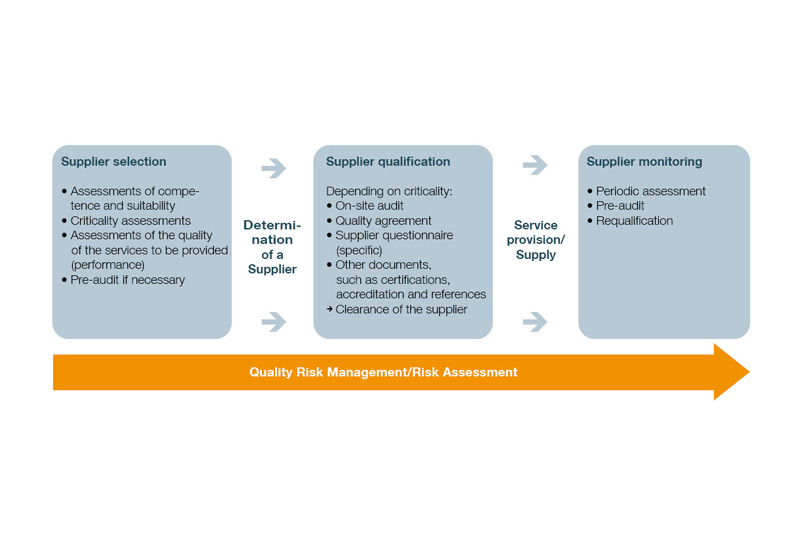
In practice, supplier management can be structured in several stages and include the following steps:
- Create evaluation catalog for supplier selection
- Carry out supplier qualification
- Select suppliers
- Establish supplier monitoring
- Supplier selection
- Supplier monitoring
Outsourced processes of medical technology